Research Highlights

Insights into bond strain in cluster-mediated synthesis
 Highlighted from cover article: John B. Parise, Bingying Xia, Jack W. Simonson, William
R. Woerne, Anna M. Plonka, Brian L. Phillips and Lars Ehm Crystals 2019, 9(5),246;
https://doi.org/10.3390/cryst9050246
Highlighted from cover article: John B. Parise, Bingying Xia, Jack W. Simonson, William
R. Woerne, Anna M. Plonka, Brian L. Phillips and Lars Ehm Crystals 2019, 9(5),246;
https://doi.org/10.3390/cryst9050246
Scientific Achievement
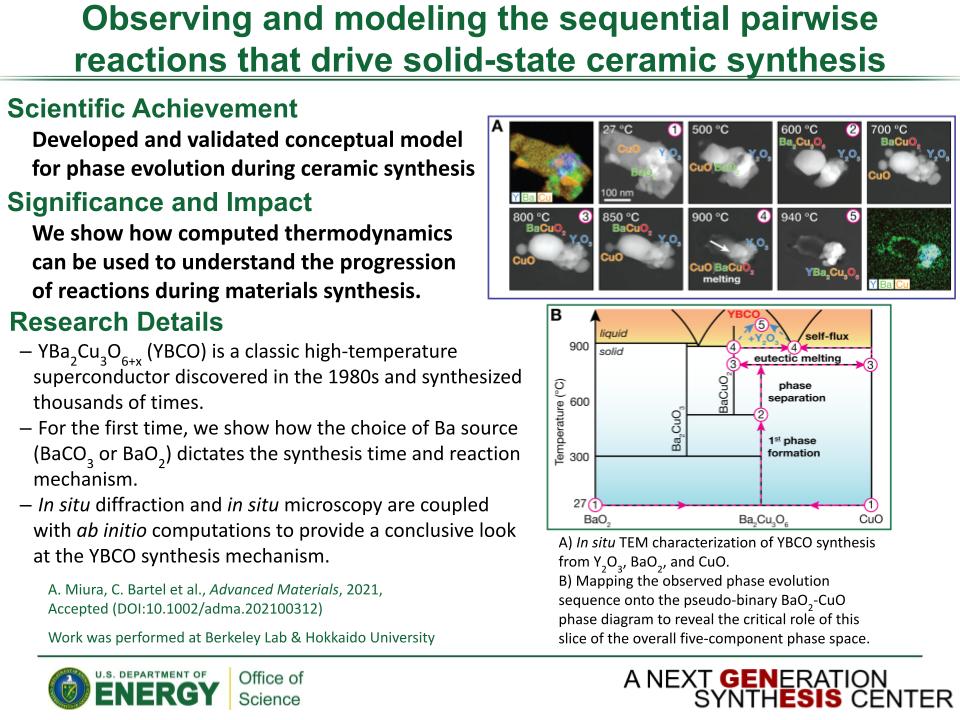
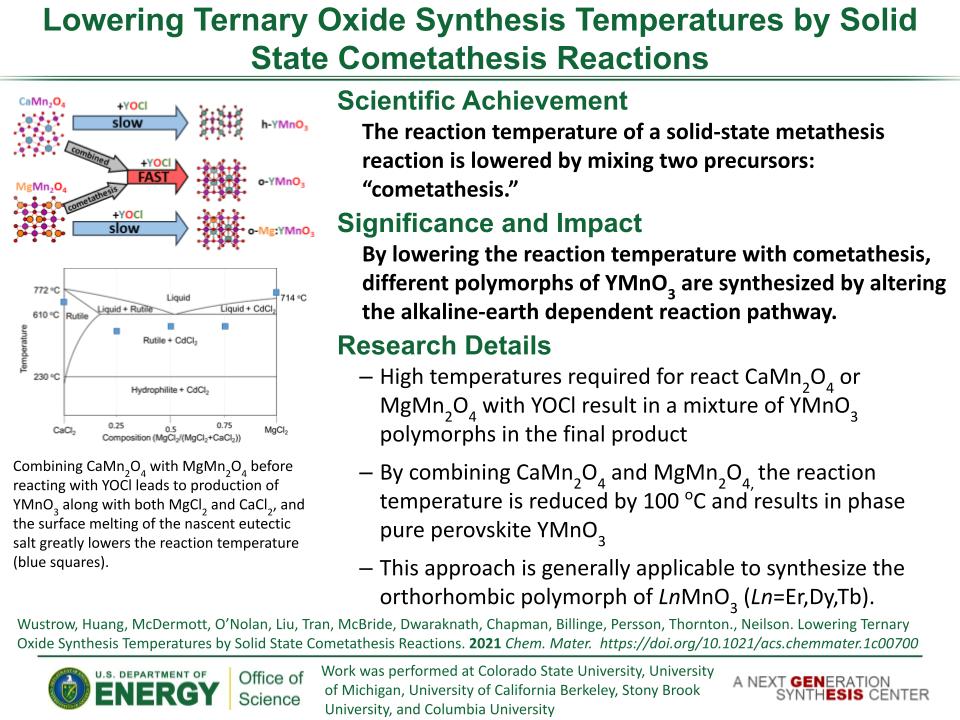
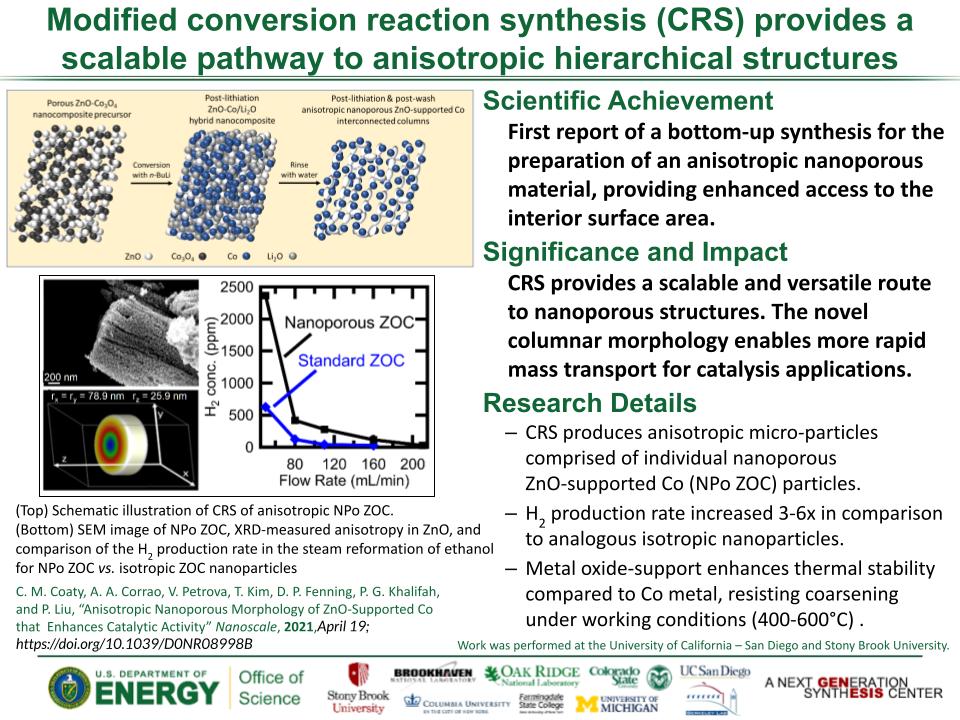
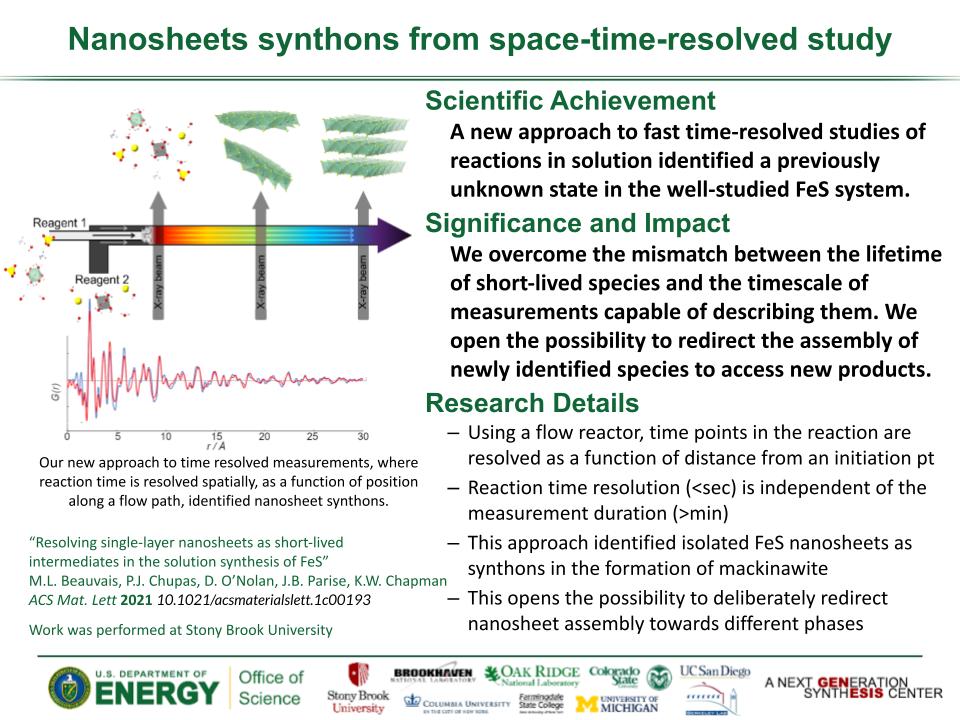
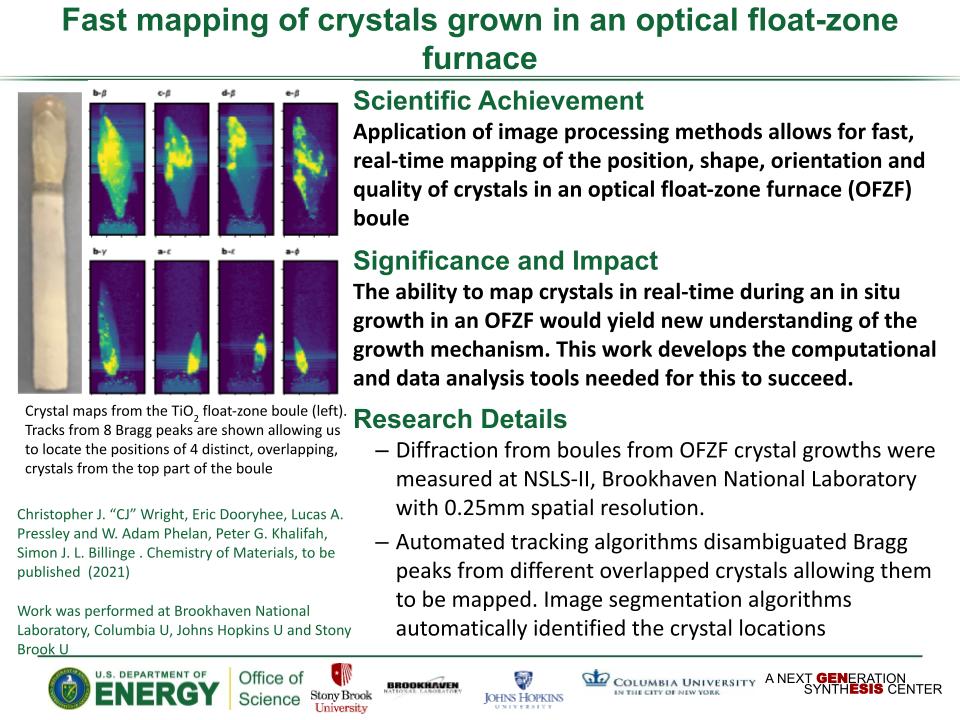
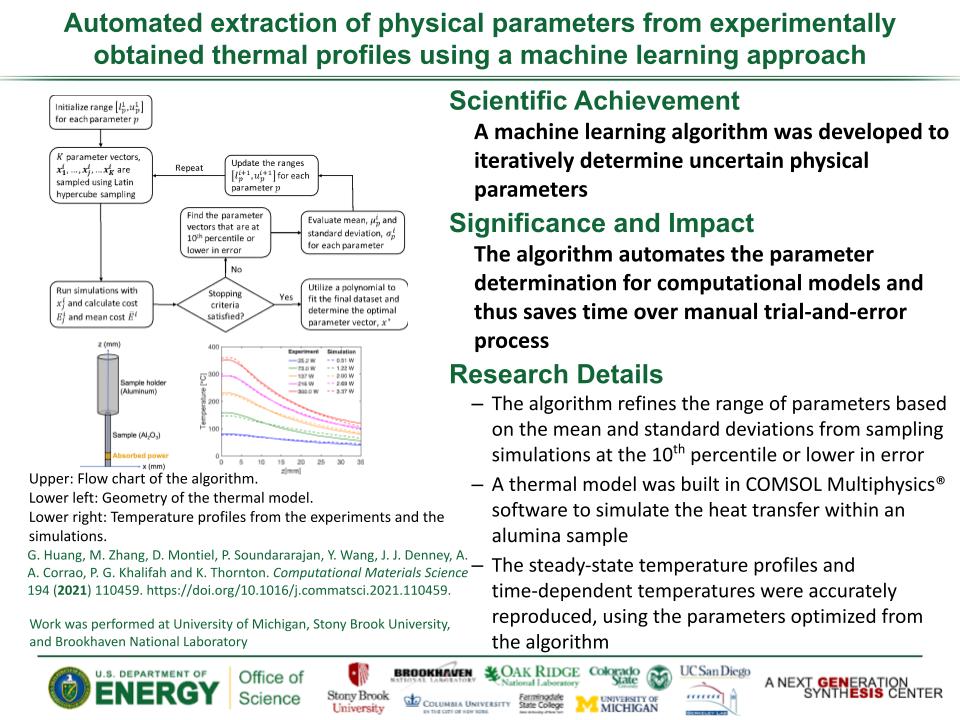
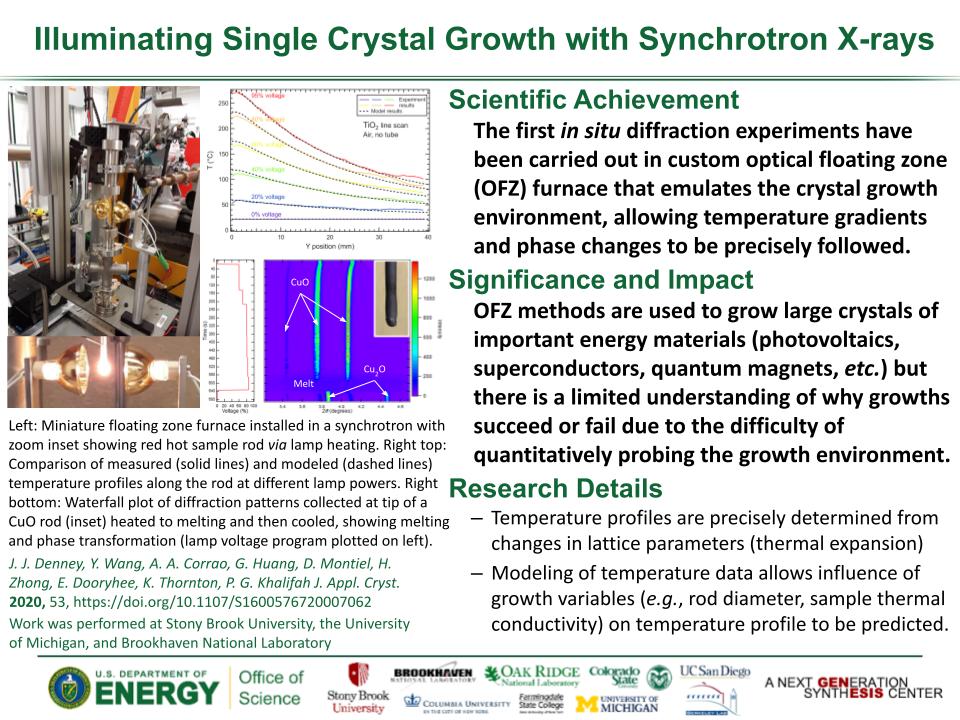
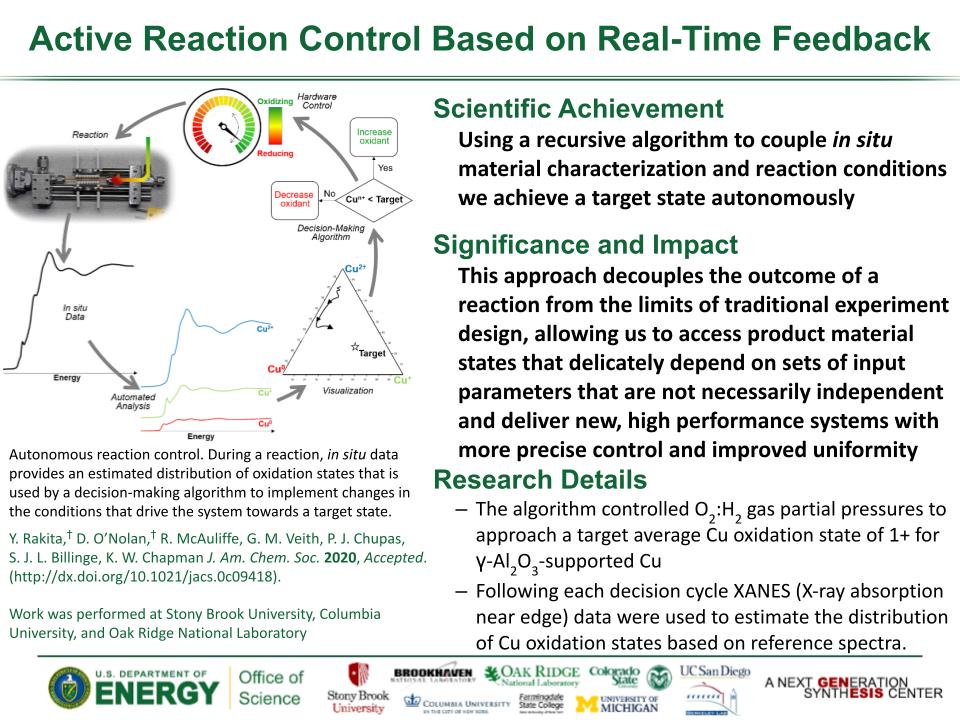
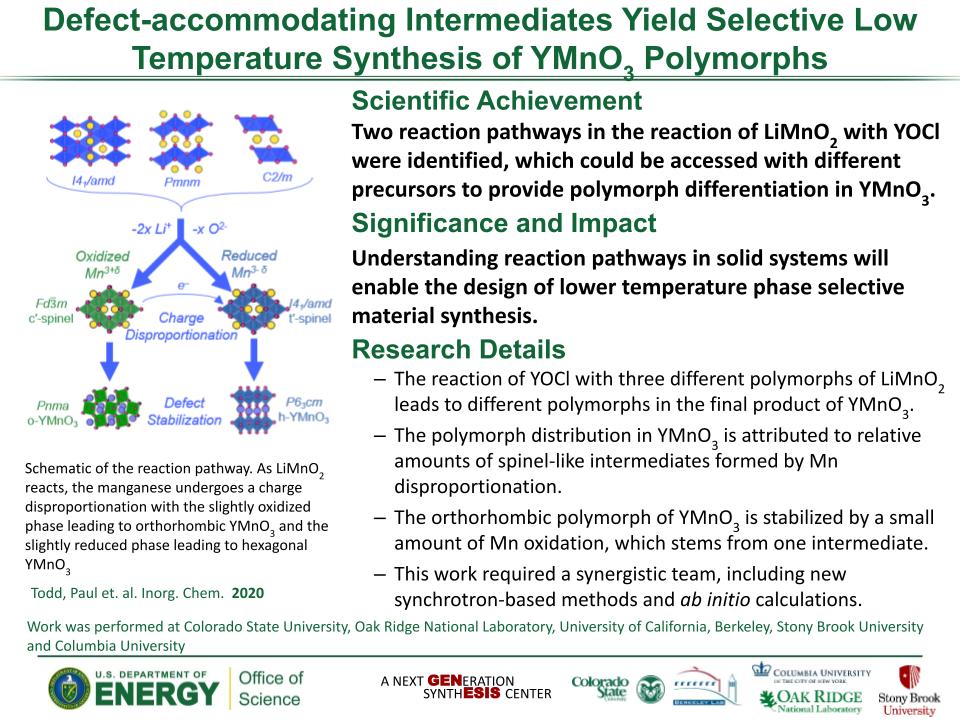
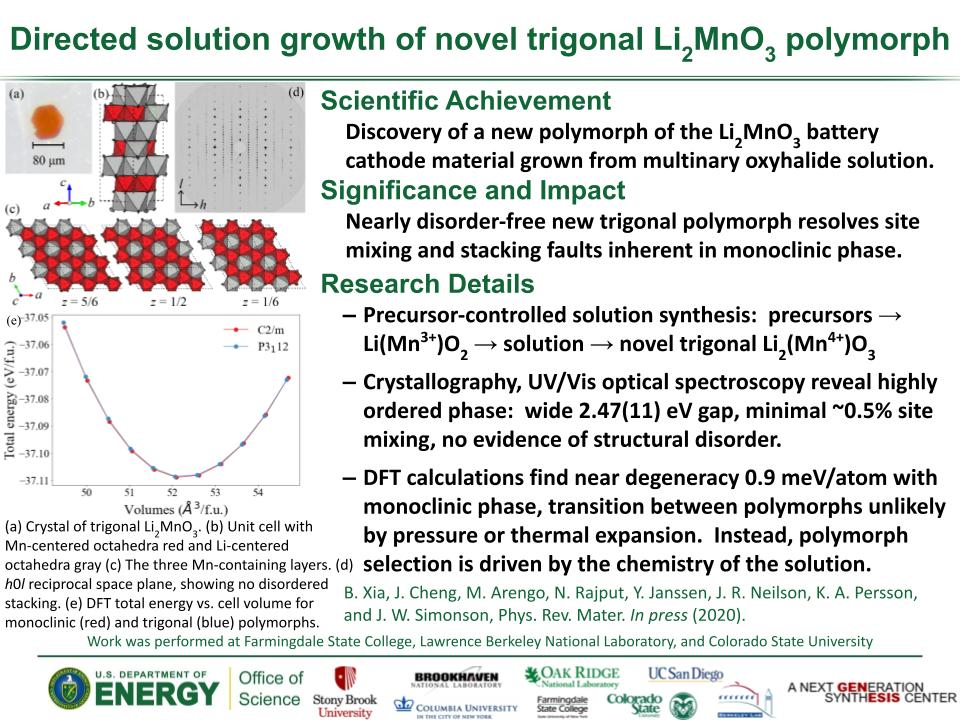
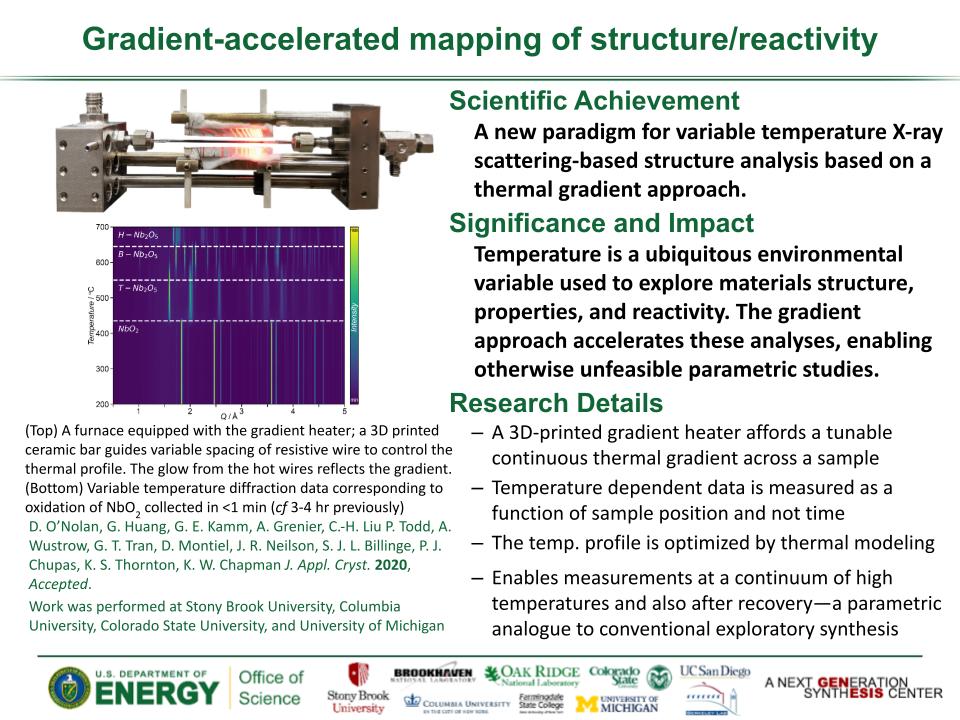
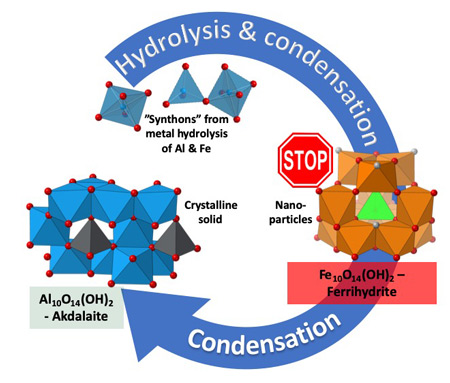
Determined the complete atomic arrangement in akdalaite, the presumed analogue of ferrihydrite, a sink for toxins in nature, and used as a synthetic barrier to contamination for environmental clean-up.
Significance and Impact
Provides insights into how the Al-analogue, which is defect free in nano- and fully condensed forms, can provide a basis to model the defect structure of ferrihydrite, which changes along the reaction pathway.
Research Details
In oxide-hydroxides condensed from clusters, significant strain accompanies conversion from cluster to extended nano-particles and crystals.
Joint x-ray, neutron and NMR studies reveal the extent of structural distortion and bond strain, compared to the clusters from which the extended solids are synthesized.
The clusters are unstrained; condensation leads to significant under-bonding of the metals in the central tetrahedral site.






