Binary Systems
1. ZnO nanowires, CuO nanowires, and α-Fe2O3 nanotubes as well as their corresponding arrays have been successfully synthesized via a low cost, generalizable, and simplistic template method. Diameters of one-dimensional (1-D) metal oxide nanostructures (~60 – 260 nm), measuring microns in length, can be reliably and reproducibly controlled by the template pore channel dimensions. Associated vertically aligned arrays have been attached to the surfaces of a number of geometrically significant substrates, such as curved plastic and glass rod motifs. The methodology reported herein relies on the initial formation of an insoluble metal hydroxide precursor, resulting from the reaction of the corresponding metal solution and sodium hydroxide, and its subsequent transformation under mild conditions into the desired metal oxide nanostructures. Size- and shape-dependent optical, magnetic, and catalytic properties of as-prepared 1-D metal oxides were investigated and noted to be mainly comparable or better than the associated properties of the corresponding bulk oxides. Ref.: ACS Nano, v.2, 944 (2008).
2. VO2 nanorods have been initially generated as reactive nanoscale precursors to their subsequent conversion to large quantities of pure, highly crystalline V2O3 with no detectable impurities. Structural changes in VO2, associated with the metallic-to-insulating transition from the monoclinic form to the rutile form, have been investigated and confirmed using X-ray diffraction and synchrotron data, showing that the structural transition is reversible and occurs at around 63ºC. When this VO2 one-dimensional sample was subsequently heated to 800ºC in a reducing atmosphere, it was successfully transformed into V2O3 with effective retention of its nanorod morphology. We have also collected magnetic and transport data on these systems that are comparable to bulk behavior and consistent with trends observed in previous experiments. Ref.: Phys. Chem. Chem. Phys., v.11, 3718 (2009) [invited].
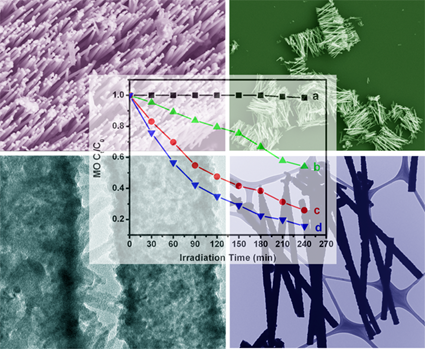
3. We have characterized (a) discrete, individual motifs and (b) arrays of crystalline and pure semiconducting metal sulfide (CuS, PbS, and CdS) nanowires, synthesized via an inexpensive, generalizable, simplistic, and ambient modified template technique. We have demonstrated control over the diameters and lengths of our one-dimensional (1-D) nanostructures through corresponding variations in the template membrane’s pore size and thickness. We have not only successfully generated cubic-phase 1-D CdS nanowires but also produced, at slightly elevated temperatures, unusual CdS cactus-like, hierarchical nanostructures, consisting of tiny nano-needles projecting out from the outer surfaces of parent CdS nanotube motifs. Vibrational properties of all of these metal sulfide nanomaterials have been extensively studied. In addition, our results indicate that our as-prepared hexagonal-phase CdS cactus-like nanotubes evinced a higher photocatalytic degradation activity than that of both cubic CdS nanowires and their commercial bulk counterparts. Ref.: Chem. Mater., v.21, 4541 (2009).
4. Herein, we report the synthesis of one-dimensional chromium oxide nanostructures, utilizing a modified sol−gel technique combined with a constrained template environment. Using scanning electron microscopy (SEM), transmission electron microscopy (TEM), and high-resolution transmission electron microscopy (HRTEM), we noted that individual nanowires were cylindrical in nature and appeared to be composed of smaller, crystalline, constituent nanoparticles, sintered and aggregated together so as to form a discrete, polycrystalline structure. Spectroscopic and diffraction investigations of our nanostructures confirmed their chemical composition with little if any impurities. Moreover, we further investigated the properties of our nanomaterials using both electrical and magnetic characterization. Interestingly, the magnetic properties of our nanostructures are strongly modified as compared with the bulk, due to the emergence of a net magnetic moment induced by uncompensated surface spins. Catalysis data showed that these nanostructures were active toward the thermal decomposition of KClO4. Ref.: Chem. Mater., v.23, 1000 (2011).
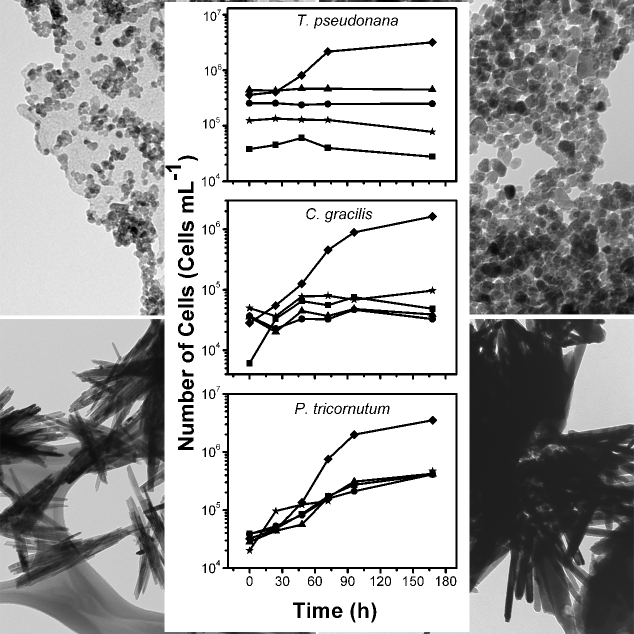
5. The influence of ZnO nanoparticle morphology on its toxicity for marine diatoms was evaluated. Four ZnO nanoparticle motifs, possessing distinctive sizes and shapes, were synthesized without adding surfactants. Diameters of ZnO spheres ranged from 6.3 nm to 15.7 nm, and lengths of rod-shaped particles were 242 nm to 862 nm. Their effects on the growth of the marine diatoms, Thalassiosira pseudonana, Chaetoceros gracilis, and Phaeodactylum tricornutum, were determined in laboratory cultures. Between 4.1 and 4.9% of the Zn from all types of nanoparticles dissolved within 72 h and was neither concentration dependent nor morphology dependent. Addition of all nanoparticles at all concentrations tested stopped growth of pseudonana and C. gracilis, whereas P. tricornutum was the least sensitive, with its growth rate inversely proportional to nanoparticle concentration. Bioaccumulation of Zn released from nanoparticles in T. pseudonana was sufficient to kill this diatom. The toxicity of rod-shaped particles to P. triocornutum was noted to be greater than that of the spheres. The overall results suggest that toxicity studies assessing the effects of nanoparticles on aquatic organisms need to consider both the dissolution of these particles and the cellular interaction of nanoparticle aggregates. Ref.: Aquatic Toxicology, v.102, 186-196 (2011).
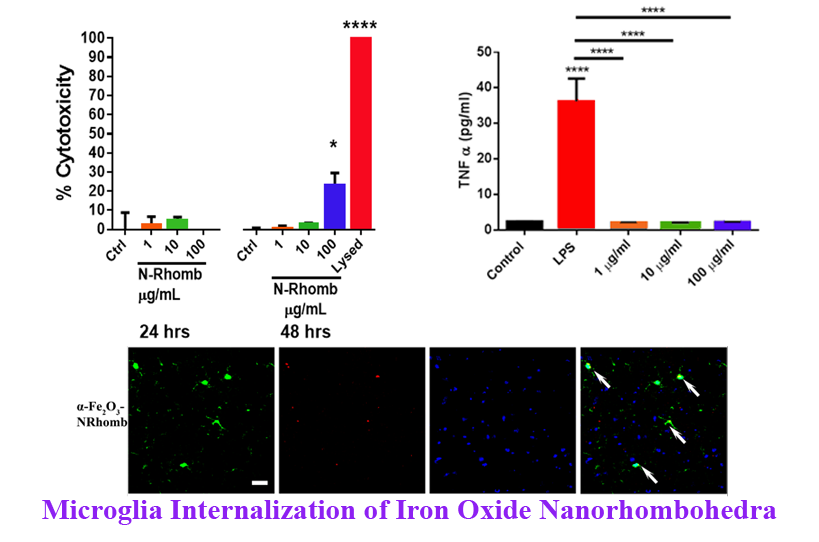
6. Understanding the nature of interactions between nanomaterials, such as commercially ubiquitous hematite (α-Fe2O3)Nanorhombohedra (N-Rhomb) and biological systems is of critical importance for gaining insight into the practical applicability of nanomaterials. Microglia represent the first line of defense in the central nervous system (CNS) during severe injury or disease such as Parkinson’s and Alzheimer’s disease as illustrative examples. Hence, to analyze the potential cytotoxic effect of nanorhombohedra exposure in the presence of microglia, we have synthesized Rhodamine B (RhB) labeled-α-Fe2O3 N-Rhomb, with lengths of 47 ± 10 nm and widths of 35 ± 8 nm. Internalization of RhB labeled-α-Fe2O3 N-Rhomb by microglia in the mouse brain was observed, and a dose-dependent increase in the cellular iron content as probed by cellular fluorescence was detected in cultured microglia after nanoparticle exposure. The cells maintained clear functional viability, exhibiting little to no cytotoxic effects after 24 and 48 hours at acceptable, physiological concentrations. Importantly, the nanoparticle exposure did not induce microglial cells to produce either tumor necrosis factor alpha (TNFα) or interleukin 1-beta (IL1β), two pro-inflammatory cytokines, nor did exposure induce the production of nitrites and reactive oxygen species (ROS), which are common indicators for the onset of inflammation. Finally, we propose that under the conditions of our experiments, i.e. in the presence of RhB labeled-α-Fe2O3 N-Rhomb maintaining concentrations of up to 100 µg/mL after 48 hours of incubation, the in vitroand in vivo internalization of RhB labeled-α-Fe2O3 N-Rhomb are likely to be clathrin-dependent, which represents a conventional mechanistic uptake route for most cells. Given the crucial role that microglia play in many neurological disorders, understanding the potential cytotoxic effects of these nanostructures is of fundamental importance if they are to be used in a therapeutic setting. Ref.: Toxicology Research, v.5, 836-847 (2016).
|