Magnetic Graphene Oxide and Graphene Nanoribbon Nanoparticles
Background
MRI contrast agents are routinely employed during MRI examinations to improve diagnostic confidence. Gadolinium (Gd)-based MRI-CAs dominate the market and are considered the “gold standard.” However, in patients with advanced renal failure, some of of these Gd-based CAs have induced a disease known as nephrogenic systemic fibrosis (NSF); a rare but severely debilitating condition that hardens and thickens the skin that results in joint contractures, restricted joint mobility and extreme pain. Furthermore, the heart, lungs and liver can be affected by Gd CAs sometimes resulting in death. Consequently, FDA has restricted their clinical use of all Gadolinium based CA in patients with advanced renal failure.
Technology
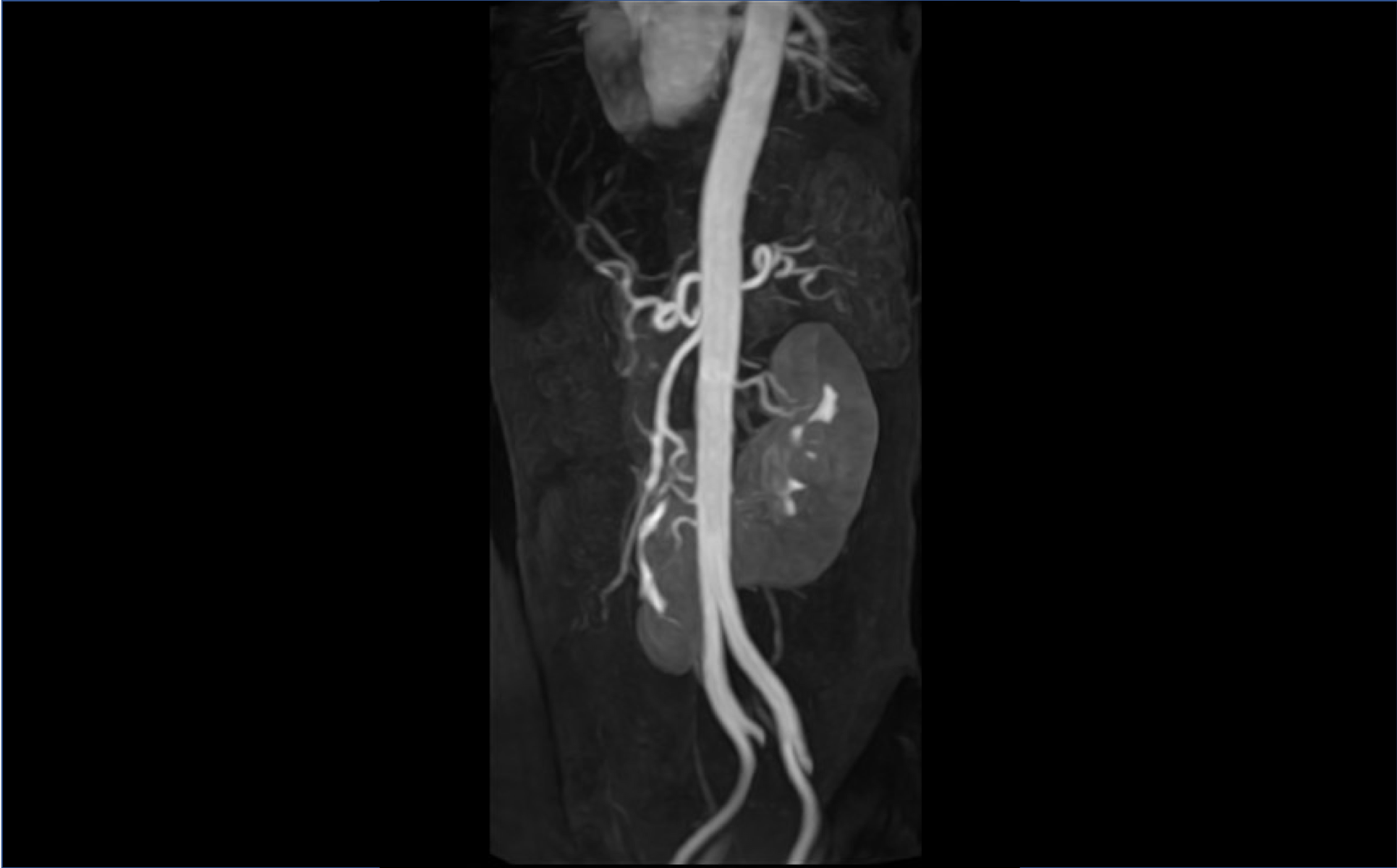
Researchers at Stony Brook University have developed a high performance nanoparticle based MRI CA consisting out of high quality graphene nanoparticles intercalated and coordinated with trace amounts of manganese. in‑vivo efficacy studies indicate that equivalent MRI performance can be achieved at substantially lower dosages than currently used CAs. This novel CA has also shown a good safety pharmacology profile including no nephrotoxicity at therapeutic doses and increased in-vivo circulation time, suggesting the possibility for its use as organ‑specific CA and an extended-residence –intravascular blood pool agent to image renal arteries.
Advantages
Easily scalable, efficient, high yielding, and amenable to suitable chemical functionalization for incorporation of other important metal atoms. High potential for development of multimodal imaging contrast agents for medical diagnostic modalities.
Application
- MRI contrast agents; - Contrast agents for existing and newly emerging multi modal imaging technologies as well as experimental forms of multi modal imaging.
Inventors
Balaji Sitharaman, Assistant Professor, Biomedical Engineering
Bhavna Paratala, Graduate Student, Biomedical Engineering
Licensing Potential
Licensing,Development partner,Commercial partner
Licensing Status
Available for license. Stony Brook University seek to develop and commercialize, by an exclusive or non-exclusive license agreement and/or sponsored research, with a company active in the area.
Licensing Contact
Valery Matthys, Licensing Associate, Intellectual Property Partners, valery.matthys@stonybrook.edu,
Patent Status
Patent application submitted
Stage of Development
Preclinical: PCT Publication No. WO 2012-154677. US9,713,650; CN201280033576.7; JP6139511
Tech ID
8288
