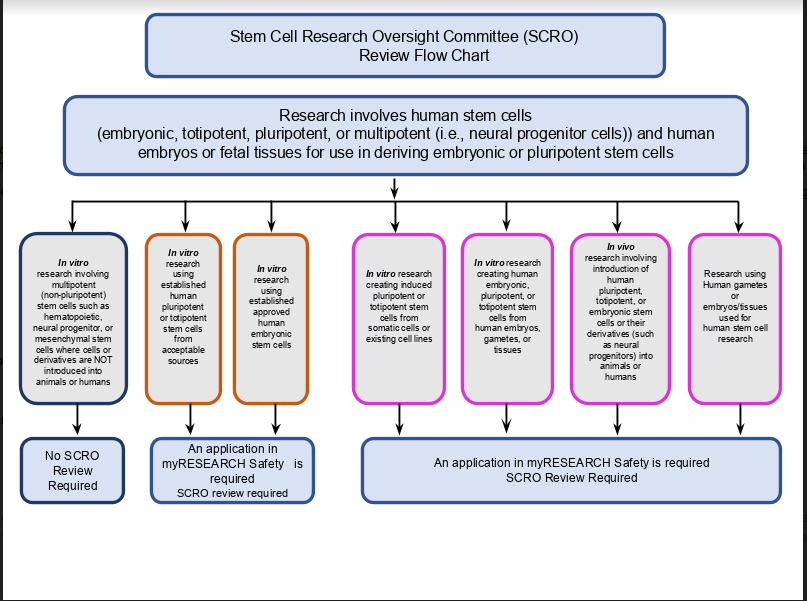
Stem Cell Use
SCRO Submission Guidelines
Complete and submit the myResearch Safety application.
See below for Instructions and Tips on completing the myResearch submission.
myResearch Safety Instructions for Initial Submission of Stem Cell Research (1/2026)
Tips for Completing the Stem Cell Research Application (SCRO) (3/2026)
In order for you to be certified to conduct research that involve stem cells, you must:
-
Complete the Stem Cell Modules in CITI
Note: Before conducting your project, you must also secure approval from all relevant oversight committees; SCRO review does not preclude the necessity of review by other research oversight committees as applicable, e.g.; Stony Brook Institutional Review Board, the Institutional Animal Care and Use Committee (IACUC), Institutional Biosafety Committee (IBC), etc.
Meetings are held as needed. No specified date.
https://stemcells.nih.gov/research-policy-and-faqs
National Institutes of Health: Stem Cell Information
https://stemcells.nih.gov/
National Institutes of Health: Stem Cell Information Policy
NIH Guidelines for Human Stem Cell Research | STEM Cell Information
National Institutes of Health: Stem Cell Research Frequently Asked Questions
NIH Guidelines for Human Stem Cell Research FAQs | STEM Cell Information
National Academy of Sciences Guidelines and Publications
https://www.nap.edu/catalog/12923/final-report-of-the-national-academies-human-embryonic-stem-cell-research-advisory-committee-and-2010-amendments-to-the-national-academies-guidelines-for-human-embryonic-stem-cell-research
UNYSTEM New York State Stem Cell Science Policy on Human Stem Cells
| Name | Department |
|---|---|
| Flaminia Talos |
Pathology and Urology |
| Soosan Ghazizadeh | Oral Biology and Pathology |
| Aaron Muth | Community Member |
| Marcia Simon | Oral Biology and Pathology |
| Bernadette Holdener, Chair | Biochemistry and Cell Biology |