Current YearTrainees
 Madeeha I. Ali
Madeeha I. Ali
B.S. Chemistry, Stony Brook University
Program/Dates Supported by NIGMS: Chemistry/ August 2024 - present
Title: Timing the Kinome: Kinetic Selectivity of Kinase Inhibitors
Kinases are key modulators of cellular signaling with significant implications in oncology, making them among the most critical drug targets of the 21st century. The development of selective kinase inhibitors is a major challenge given the high structural similarity within the kinome. Most drug discovery programs select and optimize drug leads based on thermodynamic selectivity metrics such as IC50 and Kd. This approach often overlooks kinetic selectivity, which is defined as the ability of a drug to remain bound to its intended target while rapidly dissociating from off-targets. Leveraging kinetic selectivity may improve drug efficacy and reduce side effects. My project aims to explore the utility of kinetic selectivity in developing selective kinase inhibitors. The first target is Bruton's Tyrosine Kinase (BTK), which has been validated in treating B-cell malignancies with four FDA-approved BTK inhibitors and many more in clinical trials. However, selectivity issues have raised safety concerns about liver and cardiovascular damage. I am employing Promega's NanoBRET technology to investigate kinetic selectivity in live cells, offering a measure of selectivity that is more physiologically relevant than traditional biochemical methods using purified proteins, due to its ability to monitor interactions in real-time (Figure 1). These experiments will provide insight into drug-target interactions to assess kinetic selectivity of the inhibitors by comparing their dissociation rates for the intended target i.e., BTK versus potential off-targets, such as CSK, SYK, FRK, and LYN. Furthermore, I am developing a mechanistic model that incorporates kinetic parameters such as on- and off-rates to mathematically model the intracellular drug-target binding kinetics, predicting drug behavior in more physiologically relevant conditions. Finally, cryo-electron microscopy (CryoEM) will be used to elucidate the molecular mechanisms underlying the kinetic selectivity observed
in BTK inhibitors, potentially revealing conformational changes upon inhibitor binding that drive selectivity.

Figure 1. Schematic diagram of the NanoBRET target engagement intracellular assay for binding kinetics (Figure adapted from Promega).
 Lily Cam
Lily Cam
B.S. in Biochemistry, Stony Brook University, Cum Laude
Program/Dates Supported by NIGMS: Chemistry/ June 2023-present
Title: Characterization of the heme sensing properties and the effect of heme assimilation on NosP in Pseudomonas aeruginosa
Assimilation of exogenous heme is compulsory for the prosperity of Pseudomonas aeruginosa, as well as other species of bacteria. In P. aeruginosa, two heme assimilation systems are essential - the Heme Assimilation System (Has) andPseudomonasHeme Utilization (Phu). HasA is a secreted hemophore that binds extracellular heme and brings it to HasR, a membrane receptor protein that incorporates heme into the cell. In parallel, PhuR is another membrane receptor protein that binds heme and brings it into the cell. Once heme gets internalized, PhuS then shuttles the assimilated heme to HemO, which is a heme oxygenase that breaks down heme into beta-biliverdin, carbon monoxide, and free iron that can be used for a multitude of intracellular processes. Therefore, heme assimilation systems serve as useful strategies for iron acquisition and have been proved to play essential roles in chronic P. aeruginosa infections in cystic fibrosis patients. The Boon lab has shown that NosP is a hemoprotein that senses environmental nitric oxide and labile heme to regulate the activity of a N osP-associated histidine kinase (NahK). In P. aeruginosa, our lab has shown that NosP/NahK signaling pathway is essential for sensing NO to trigger biofilm dispersal; however, it is still unknown if NosP/NahK signaling pathway also possesses additional functions of sensing assimilated heme to elicit downstream physiological responses, and whether the heme sensing properties of NosP/NahK are functionally dependent on Has and Phu systems.
Pyocyanin is a blue colored redox active phenazine, a major virulence factor. Its production is positively regulated by pseudomonas quorum sensing molecule, PQS. This can be correlated to data from our laboratory in PA14; when nahK is knocked out, an increase in pyocyanin and PQS levels are also observed. Since ∆nahK gives a similar phenotype to ∆ hemO, it is suggestive that the heme assimilation systems and/or HemO activity may be epistatic of the NosP/NahK signaling system. Heme assimilation is required by P. aeruginosa in chronic infections, therefore understanding and ultimately targeting heme sensing/assimilation pathways may help combat chronic pseudomonas infections.
 Emma Gebauer
Emma Gebauer
BS Biochemistry & Cell Biology, minor in Physics Bucknell University, 2020
Program/Dates Supported by NIGMS: Chemistry/ August 2024 - present
Title: Structure-Guided Design of Isoform-Specific Cyclophilin Inhibitors
Strokes are one of the leading causes of death worldwide, and ischemic strokes account for about 87% of strokes in the US and about 62% of strokes globally. Ischemic strokes are made more complicated by ischemic-reperfusion injury (IRI), in which cells begin to rapidly dysfunction and die during the restoration of blood flow to the ischemic tissue. In the heart and brain, IRI is exacerbated by dysregulation of the mitochondrial transition pore (mPTP). Cyclophilin D (CypD) is a peptidylprolyl isomerase and is a key modulator of the mPTP, where binding of CypD to the mPTP favors the open conformation of the pore. Inhibition of CypD lowers the probability of pore opening when there is substantial inorganic phosphate, Pi, present, suggesting CypD inhibition is a viable therapeutic target.
Currently, there is only one FDA-approved cyclophilin inhibitor, Cyclosporine A (CsA), which, while potent, is non-specific and binds to several cyclophilin isoforms, leading to unwanted side effects. Our group has previously shown that building compounds into the specificity pocket of the CypD active site is a promising approach to developing isoform-specific inhibitors, though these initial compounds had poor cell permeability.
To address this, we are using a fragment-based drug design approach in combination with computational docking to identify novel CypD inhibitors with more desirable small molecule chemistries. We then validate the computational results with traditional biochemical and structural biological approaches. .
 Karishma Ramcharan
Karishma Ramcharan
M.S. in Chemistry, Stony Brook University, 2023
B.S. in Biochemistry, Hofstra University, 2021
Program/Dates Supported by NIGMS: Biochemistry/ August 2024 - present
Title: Regulation of the T3SS and T6SS through the NahK/HptB phosphorelay signal in Pseudomonas aeruginosa
Pseudomonas aeruginosa (P. aeruginosa) is a Gram-negative, opportunistic pathogen known for causing nosocomial infections, such as lung infections of cystic fibrosis patients, ventilator-associated pneumonia, catheter-based infections, and topical infections of burn victims and surgery patients. The pathogenicity of P. aeruginosa stems from the bacteria’s ability to form biofilms, allowing the bacteria to survive in the presence of host immune responses. Biofilms are multicellular communities formed when bacteria use extracellular appendages to attach to a surface and self-encapsulate within a thick matrix called the extracellular polymeric substance (EPS). The transition from a planktonic, motile state to a sessile state is regulated by multiple signal transduction systems that modulate intracellular levels of the signaling molecule cyclic-di-GMP. This second messenger functions as a cofactor for proteins involved in biofilm-related gene transcription. As P. aeruginosa is becoming progressively resistant to current antibiotics and treatment regimens due to biofilm formation, there is an increasing need for developing new and alternative methods of treating infections caused by this pathogen. Investigations into biofilm dispersal have shown promising results that, if characterized correctly, could improve the treatment of these infections.
P. aeruginosa secretes many virulence factors through secretion systems to cause colonization, cytotoxicity, and evasion of an innate host immune system. This pathogen utilizes Type III Secretion Systems(T3SS) and Type VI Secretion Systems(T6SS), which function antagonistically to control the switch between acute-to-chronic infections, respectively. Our lab has established phenotypic characterizations in the NahK/HptB multikinase network to support the current findings that NahK is a central regulator of biofilm formation through the HptB/RsmA system, however our data also shows that some of the phenotypes observed, such as increased intracellular c-di-GMP, EPS production, and increased transcriptional expression of T3SS, function independently of RsmA through unknown regulatory mechanisms. This project aims to investigate how the phosphorylation from NahK to HptB can regulate the production of intracellular c-di-GMP and the observed phenotypes through a RsmA-independent pathway. We also aim to probe the characterized cyclic-di-GMP regulating enzymes, known to directly modulate the HptB network, to better understand the role of cyclic-di-GMP in the transcription of secretion systems.
 Hayden Snyders
Hayden Snyders
B.S. in Chemistry, Saint Louis University, summa cum laude
Program/Dates Supported by NIGMS: Chemistry/ August 2023-present
Title: Exposing the mechanistic nature of Alzheimer’s disease: Development of a self-consistent coarse-grained model for tau protein simulation
Affecting more than 5.6 million people in the United States in 2020 and doubling in cases every 5 years for people over the age of 65, Alzheimer’s disease (AD) is one of several neurodegenerative diseases associated with the dysfunction of tau protein. In the central nervous system, tau is responsible for aiding in the development and stabilization of neuronal microtubules, which are critical to the integrity and function of axons. In AD and other forms of dementia, tau protein aggregates irreversibly in the presence of RNA and other biomolecules resulting in the deposition of “tau tangles”, a process strongly correlated with disease progression. The mechanism by which this aggregation occurs remains poorly understood, impeding the development of therapeutic remedies for AD and other tauopathies.
Using light scattering experiments and coarse-grained molecular dynamics simulations, my project focuses on developing a self-consistent model for predicting the structural and thermodynamic properties of tau protein in solution. Taking into consideration the interplay between electrostatic, hydrophobic, and hydrogen bonding interactions, the model will predict tau behavior based on its primary sequence of amino acids and their associated side-chain bonding properties, such as their charges at physiological pH. Elucidating the effect of intermolecular interactions on the properties of tau protein, this model will provide a strong foundation for understanding greater mechanisms of aggregation, an important step towards the development of therapeutic agents for AD and other tauopathies.
 Grace Yoon
Grace Yoon
B.S. in Chemistry, Stony Brook University
Program/Dates Supported by NIGMS: Chemistry/ June 2023-present
Bruton's Tyrosine Kinase (BTK) is a cytoplasmic tyrosine kinase that serves as a promising therapeutic target for various B-cell malignancies, including Mantle Cell Lymphoma (MCL), Chronic Lymphocytic Leukemia (CLL), and Diffuse Large B-cell Lymphoma (DLBCL). Extensive research has been conducted to develop effective BTK inhibitors, but these often lead to undesirable side effects and drug resistance. To overcome these limitations, a new approach using proteolysis targeting chimeras (PROTACs) has emerged, aiming to degrade BTK instead of solely inhibiting it. The Tonge group has successfully developed a novel BTK PROTAC named PTD10; however, further improvements are required to enhance its degradation ability and potency. Moreover, several unanswered questions remain regarding the mechanism of action of this degrader, particularly in terms of its kinetics. Therefore, my project will primarily focus on the development of a second-generation BTK PROTAC and the investigation of crucial kinetics properties, including binding kinetics and the rate of ubiquitination of the degrader.
Active Trainees
 Juan Alcantara
Juan Alcantara
Skidmore College, Chemistry B.A., Biochemistry Concentration, Summa Cum Laude
Program/Dates Supported by NIGMS: Chemistry/ 2022-2024
Title: Rapid rescoring and refinement of ligand-receptor complexes using mixed MD/MC with a pose reservoir
Modern drug discovery approaches commonly utilize computational methods to find or generate molecules that show favorable binding to a specified macromolecular target. Millions of molecules can be rapidly tested via a virtual screen to filter out bad binders early before purchasing or synthesizing the molecules for experimental assays. Virtual screening involves the generation of poses for a library of ligands, and ranking using simple energy functions with limited flexibility and generally, no water solvation. Top scored poses are used to rank and prioritize ligands with respect to their theoretical interaction energies with the target. The simplified electrostatics approximations and lack of solvation limit the accuracy of the virtual screening pose energy rankings, increasing incidents of choosing the incorrect pose as the most favorable one. Here we adapt an accelerated molecular dynamics (MD) method, developed for peptide folding, to enhance and refine the virtual screening pose energy ranking process. The method re-ranks the poses using the full molecular mechanics energy function and allows for receptor flexibility as well as solvation, increasing the accuracy of the predicted pose rankings relative to the simplified virtual screening protocols. ligand poses from the virtual screen are subjected to short MD equilibration and are used to populate a structure reservoir which then can be used alongside another MD simulation of the poses to allow for rapid exchanges between the conformations via Monte Carlo (MC) jumps. This allows us to circumvent the sampling time constraint of normal MD which would be too slow in generating the same poses that were already predicted by the virtual screen. Relative populations of poses can be obtained with regards to their binding interaction stability. This method provides a more accurate way to rank ligand poses to either corroborate or refine the original virtual screening rankings.
Recently we have shown that this new MD approach with implicit solvation can predict the most favorable pose amongst the ones generated in a DOCK virtual screen for systems where DOCK also chose the same pose as the favorable one. The approach was also able to select the correct pose in instances where DOCK was not able to predict it as being the most favorable. Since MD is more accurate in its sampling dynamics, the pose rankings and favorability observed during the MD simulations can be used as a guide to determine whether the initial DOCK screening was accurate or if the rankings could benefit from MD refinement. Although this approach showed success with implicit solvent, ongoing work will focus on using explicit solvation to account for systems that may have critical water molecules mediating ligand binding, as well as developing an additional approach to account for residue protonation states in cases where shifted pKa’s in the active site are ambiguous. Further in the future this MD method will be adapted for use in estimating relative binding affinities for different types of ligands rather than just different conformations of the same one. This will allow for the comparison of very distinct ligands which is not feasible with current free energy methods that rely on ligands having similar scaffolds.
 Karla Cardenas Arevalo
Karla Cardenas Arevalo
B.S. in Biology at The City College of New York CUNY
Program/Dates Supported by NIGMS: Microbiology & Immunology/ 2023
Title: Pilus biogenesis in uropathogenic E. coli (UPEC)
Urinary tract infections (UTIs) are one of the most common infectious diseases in both community and hospital settings cause primarily by the uropathogenic Escherichia coli (UPEC). Although standard antibiotic treatment is often successful in clearing UTIs there is a high rate of recurrence. In addition, there is emergence of multidrug resistant bacteria that represents a major health threat, for which new alternatives to traditional antibiotics are urgently needed. My research focuses on understanding the intricate molecular mechanisms utilized by the Gram-negative bacteria (UPEC) to coordinate the assembly and secretion of cell surface structures called pili. Pili serve as critical virulence factors, and the knowledge acquired from this research holds promise for the development of anti-virulence therapeutics. In my project, I have set out to employ the powerful technique of FRET (Förster resonance energy transfer) to probe and quantify specific interactions occurring at the bacterial outer membrane during pili assembly, with a specific focus on P pili, which play an important role in facilitating kidney colonization. I aim to acquire an understanding of how the usher selectively recognizes pilus subunits in the correct order, as well as the mechanisms underlying activation of the usher to initiate pilus biogenesis. This research project will elucidate fundamental mechanisms of protein translocation across membranes, the assembly of complex organelles, and virulence factor biogenesis at the bacterial outer membrane. Another facet of my project involves harnessing the power of FRET as a tool to screen small molecules, probing their potential to disrupt the process of pilus biogenesis. My project will facilitate the identification of novel anti-virulence compounds that inhibit assembly of adhesive pili by Gram-negative bacterial pathogens, selectively disrupting host-pathogen interactions required to initiate and sustain infection.
 Kindra Becker
Kindra Becker
B.A. in English Literature, Master of Library and Information Science, University of South Carolina
Program/Dates Supported by NIGMS: Chemistry/ 2022- 2024
Title: Synthesis of Fluorophores for APEX2 Proximity Labeling in Mycobacterium tuberculosis
Mycobacterium tuberculosis (Mtb), the causative agent of tuberculosis (TB), is the second leading infectious killer after COVID-19. The rise of multi-drug- and extensively drug- resistant cases of TB reveal a pressing need for the development of new drugs against Mtb. Although an attractive drug target is the unique diderm envelope that harbors essential processes like uptake and export of metabolites, the mycobacterial cell wall proteome is not well-elucidated. In determining the proteome of the cell wall, we can identify these essential processes and, within these, potential drug targets. Our lab has successfully used proximity labeling with APEX2, an engineered peroxidase, to label proteins in a compartment-specific manner. To expand our understanding of the mycobacterial cell envelope, we have genetically encoded APEX2 to an outer membrane protein, MspA. Biotin-phenol, a commonly used APEX2 substrate, is cell permeable, labeling proteins in both the periplasm and cell wall. Considering this, we hope to synthesize a substrate that is unable to enter the cell. Here, we use the coupling reagent N-N’diisopropylcarbodiimide (DIC) to activate a carboxylic acid and react with a free amine to form an amide bond. 5-aminofluorescein (FAM) was chosen as the basis for the cell impermeable substrate because of its hydrophilicity and anionic charge at neutral pH. As a control, the starting material 7-hydroxycoumarin-3-carboxylic acid was chosen for its ability to cross the mycobacterial membrane. These products, FAM-phenol and 7-hydroxycourmarin-phenol, will hopefully help us cross the final frontier to understanding uptake and export at the mycobacterial cell surface.
 John Bickel
John Bickel
BS in Chemistry, Truman State University
Program/Dates Supported by NIGMS: Applied Mathmatics/ 2020-2021
Title: Strategies for biasing de novo design in DOCK6
The goal of my proposed research is to further develop and improve the from-scratch construction algorithm (de novo design) as currently implemented in DOCK6 for the purposes of constructing potential compounds designed for a specific target. As libraries of compounds on public databases such as ZINC15 continue to grow in size at an astounding rate (currently ~750M), performing a full virtual screen of these libraries against potential drug targets is becoming increasingly infeasible given the current docking architecture. By constructing these compounds directly to the site of interest, there is a higher probability of the resulting compounds to have higher specificity and tighter binding interactions.
The ultimate goal in improving and optimizing the DOCK6 de novo algorithm is to be able to effectively bias construction of new molecules such that populations of newly constructed torsion types (allowable bond types) more closely match the distribution of torsions of an input library derived from drug-like compounds. This goal is being addressed by implementation of a fitness proportionate (roulette) method of selection, as well as implementation of new ways of handling DOCK6’s database of fragments and their associated bonding pairs.
Danielle Cervasio
BS, summa cum laude, SUNY Cortland, 2017
Program/Dates Supported by NIGMS: Chemistry/ 2018-2021
Title: Translational applications of small-molecule, astrocyte-targeted probes
Astrocytes are the most abundant non-neuronal cell type in the brain, providing support, nutrient buffering, and instructions to neighboring cells, particularly neurons, in order to maintain normal physiological function and homeostasis. Importantly, astrocytes play a large role in the brain by responding to insult, communicating with neurons via gliotransmitters, and forming protective barriers around endothelial cell layers. Because astrocytes are present abundantly in all cortical areas of the brain, performing essential metabolic, structural, and homeostatic roles, it is not surprising that they are implicated in a slew of disease and disorders, including, but not limited to, Alzheimer’s, Parkinson’s, glioblastoma, stroke, seizure, infection, and depression. Thus, it is becoming increasingly important to consider these cells for therapeutic intervention of such disease, seeing that they are important players in maintaining normal brain function. In leveraging organic cation transporters (OCTs) on the surface of astrocytes, we have developed a system by which a permanently positive N-heterocyclic amine can be attached, via organic chemistry, to diverse small molecules like fluorophores, photoactivatable markers, or calcium indicators and delivered specifically to astrocytes over neurons and other glia (Fig. 1a and b). These data have inspired us to think about more translational applications, including the potential to deliver drugs or transcription activators to astrocytes for therapeutic intervention. Gene-targeted therapies are currently in the clinic, but there exists a lack of regulated systems. We currently have some control over where, but no control over when therapeutic genes are expressed. I plan to synthesize and assay a doxycycline modified with a permanently positive N-heterocycle to exploit the powerful tetracycline-inducible gene expression system (Tet-On/Off) in order to control gene expression in astrocytes, exclusively. In adding this modification, I hypothesize that we will be able to target astrocytes for therapeutic intervention, decrease concentrations of drug needed by increasing blood brain barrier (BBB) penetration, and reduce off target effects of current constitutively active therapies as seen in Glial cell-derived neurotrophic factor (GDNF) or Neurturin (NTN) gene therapy for Parksinson’s disease.
 Christopher Corbo
Christopher Corbo
B.E. in Chemical & Molecular Engineering, Stony Brook University
Program/Dates Supported by NIGMS: Biochemistry and Structural Biology/ 2021-2023
Title: Dynamic scoring for de novo design of molecules
Computer aided drug design is an integral part of modern drug discovery, particularly for hit identification. A hit is typically defined as a molecule which has a micromolar affinity for a protein target and is a starting point for optimization. This stage of drug development is particularly amenable to the use of computational techniques as they can search large areas of chemical space in a quick and efficient manner. My research aims to further improve this process via the development of new routines and algorithms in DOCK6.
One such method for hit identification is the fragment by fragment construction of molecules within the constraints of a binding site, called de novo design. Currently DOCK6 can bias the construction of molecules towards those which are more similar to a given reference molecule, such as an experimentally validated inhibitor. This is achieved through the use of similarity-based scoring functions which assess the similarity of growing molecules to the reference molecule. Those which are most similar are advanced to subsequent stages of growth. However this inadvertently creates a bias for molecules which are bulkier at early stages of growth, and leads to ill-informed decisions. I believe that by reconstructing the reference molecule in parallel to the growth of new molecules, a higher degree of similarity can be achieved. This dynamic scoring will further focus the search for new hits in areas of chemical space which are likely to be successful, while still identifying novel chemical structures.
 Alexa Gilberti
Alexa Gilberti
BS in Chemistry, Stony Brook University, magna cum laude
Program/Dates Supported by NIGMS: Chemistry/ July 2020-2022
Title: Targeted protein Degradation of SMG-1
Serine/Theonine protein Kinase one (SMG-1) is involved in nonsense mediated mRNA decay (NMD) which is one of the DNA Damage Response (DRR) pathways in mammalian cells. The DDR serves to detect, fix, or ameliorate errors that may arise during DNA replication, and if left unchecked can lead to cell cycle arrest, regularion of DNA replication, and the failure to repair of DNA damage. DDR plays an important role in protecting cancer cells from chemotherapeutics, and the underlying hypothesis of this project is that inhibition of SMG-1 will senstive cancer cells to cancer therapies as well as the immune response. However, efforts to evaluate this hypothesis using genetic knockouts in breast cancer cells have been hindered by the essentiality of SMG-1.
Proteolysis targeting chimeras (PROTACs) utilize the cells natural ubiquitin proteasome system for protein degradation and maintaining homeostasis in the eukaryotic cells, in order to force the initiation of a degradation cascade on a target. In order to determine if SMG-1 is a suitable target for PROTAC development, we will utilize the Achilles TAG (aTAG) tool that was developed by C4 Therapeutics to knockdown the SMG-1 protein. A fusion protein of the aTAG with SMG-1 will be created using CRISPR/Cas9, and PROTACs directed at the aTAG will be used to degrade the aTAG/SMG-1 fusion protein. Targeted protein degradation with PROTACs will be used to chemically knockdown SMG-1 and evaluate SMG-1 as a potential therapeutic target.
 Nipuni Gunawardhana
Nipuni Gunawardhana
BS in Chemistry, University of Colombo, Sri Lanka
Program/Dates Supported by NIGMS: Chemistry/ 2020-2021
Title: Astrocyte Specific Delivery of Methyl-Pyridinium Tagged Small-Molecule Cargo
Neurons were the big players in neuroscience research until recent efforts identified that astrocytes too are active players in brain function. Astrocytes are the most abundant type of non-neuronal cells in the brain. In addition to maintaining blood-brain barrier integrity and providing nutrients to the surrounding cells, astrocytes form a specialized structure with pre- and post-synaptic neurons known as the tripartite synapse where they take part in the uptake and release of neurotransmitters. Astrocytes have been implicated in many neurological diseases like Parkinson’s disease (PD), Alzheimer’s disease (AD), Amyotrophic lateral sclerosis (ALS), glioblastoma and depression. Accordingly, to understand how the brain circuitry functions it is essential to study astrocytes.
However, only a limited number of strategies are available for astrocyte visualization and manipulation. These existing methods lack specificity and versatility. A new class of methyl-pyridinium tagged fluorescent markers has been developed in our lab that shows specific targeting and visualization of astrocytes. The positive charge of the methyl-pyridinium targeting moiety of this probe allows active transport of diverse cargo such as Ca2+ sensors, small molecule drugs and transcriptional activators to astrocytes through the astrocyte resident Organic Cation Transporter (OCT).
I am currently synthesizing a methyl-pyridinium tagged cyanodoxycycline to activate transcription of specific genes in astrocytes using the tetracycline-inducible gene expression (Tet-On) system. My work is also focused on the drug tamoxifen to create a traceless astrocyte delivery strategy. These molecules will help us to evaluate the ability of the targeting moiety to deliver drugs specifically to astrocytes. This strategy will be beneficial for exploring the therapeutic aspects of astrocytes in various neurological diseases and will improve our understanding of the functions of astrocytes in the brain.
 YongLe He
YongLe He
BS in Chemistry, Stony Brook University, 2019
Program/Dates Supported by NIGMS: Chemistry/ 2022-2024
Title: Mechanistic studies of BLUF and LOV photoreceptors and photoswitchable fluorescent protein
Photoreceptors are light-sensitive proteins that contain a chromophore responsible for sensing and responding to light in a variety of organisms. They have unique applications in the field of optogenetics where light can be used to modulate biological functions. My research involves the characterization of two groups of photoproteins: the blue-light using FAD (BLUF) and light oxygen voltage (LOV) domain photoreceptors where excitation results in structural changes that ultimately lead to biological processes such as photosynthesis, visual transduction, and circadian rhythms. For the BLUF and LOV proteins, our aim is to elucidate the signaling transduction mechanism from the chromophore binding domain to the effector domain using time-resolved infrared spectroscopy combined with chemical and structural biology techniques. Currently, we are investigating multiple BLUF (AppA, PixD, and BlsA) and LOV (AsLOV2, YtvA, and El222) proteins. Also, we are correlating the structural activity relationship of the photoactivated domain with the functionally relevant effector domains, such as PixD-PixE complex (controls phototaxis), AsLOV2-PAC1 (optogenetic construct), YtvA-FixL1 (histidine-phosphotransferase and Kinase), and El222-DNA complex (transcriptional factor).
In addition, we also study the photoswitchable fluorescent protein that can be reversibly switched between bright and dark states. The main goal is to characterize the photoswitching mechanism in Dronpa2 using vibrational spectroscopy (IR and Raman), isotope labeling, and unnatural amino acid incorporation. Previously, we successfully used IR and Raman spectroscopy to explain the photoswitching isomerization of the Dronpa chromophore, and we want to extend our study to its homolog Dronpa2. Ultimately, this knowledge can drive the development of more optogenetic tools and the application of photoswitchable proteins in super-resolution microscopy.
 Adeline Luperchio
Adeline Luperchio
B.S. in Microbiology, University of Vermont
Program/Dates Supported by NIGMS: Microbiology & Immunology/ 2022- 2023
Title: Critical HIV-1 Vif interactions: Molecular approaches to novel HIV therapies
Like all viruses, HIV-1 hijacks cellular machinery to facilitate replication and evade host antiviral defense mechanisms. HIV-1 deploys several accessory proteins that manipulate the cellular environment to enhance virus replication. The accessory protein Vif is responsible for counteracting host antiviral APOBEC3 enzymes to promote virus replication. In the absence of Vif, APOBEC3s package into nascent HIV-1 particles and hypermutate the viral genome to kill the virus. Vif counteracts APOBEC3 restriction by targeting them for proteasomal degradation prior to virus encapsidation. Quantitative proteomics studies of HIV-1 infected T cells recently revealed that Vif also induces remodeling of the host phosphoproteome by inactivating protein phosphatase 2A (PP2A) complexes. PP2A is responsible for the vast majority of cellular serine/threonine dephosphorylation events in most cell types. How phosphoproteome remodeling enhances HIV-1 replication has yet to be resolved.
Another recently ascribed Vif interaction is with the cellular kinase AKT. Vif stimulates hyperactivation of AKT to trigger a feedback loop wherein AKT phosphorylates Vif to enhance its stability. Interestingly, inhibition of AKT leads to a complete loss of Vif protein accumulation; however, the molecular mechanism driving this process has yet to be elucidated. My preliminary data indicates that Vif-induced inactivation of PP2A triggers hyperactivation of AKT. Thus, I’m interested in if the Vif-PP2A-AKT network can be leveraged as a therapeutic target to deplete Vif protein levels and restore antiviral activity of APOBEC3 restriction factors. The Aims of my research are to 1) use biochemical, genetic, and pharmacologic approaches to investigate the Vif-PP2A-Akt nexus and 2) identify small molecule inhibitors that disrupt the Vif-PP2A interaction. These mechanistic insights may inform the development of novel strategies that restore restriction activity of the APOBEC3 enzymes, potentially taking the next step towards curative therapeutics.
 Luz Mendez
Luz Mendez
BA with honors, Wesleyan University, 2017
Program/Dates Supported by NIGMS: Chemistry/ 2018-2021
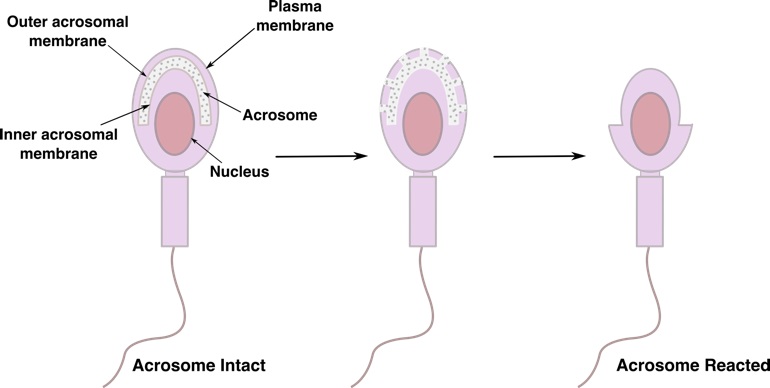
Title: Identifying Inducers of the Acrosome Reaction in Human Sperm with Synthetic Glycopolymers
The acrosome reaction (AR) in spermatozoa is an essential process in mammalian fertilization. Although the mechanism behind the activation of AR is not well-understood, it is known that the glycoproteins surrounding the egg cell’s zona pellucida (ZP) can mediate the activation of AR through a series of carbohydrate binding events with the receptors on the sperm plasma membrane. Previous research in the Sampson lab has demonstrated that AR in mouse sperm can be induced successfully in vitro with the aid of glycopolymers. These glycopolymers, synthesized by ring-opening metathesis polymerization (ROMP), possess norbornene backbones and resemble terminal residues of the sugar moieties present on the ZP. These glycopolymers are hypothesized to be potential inducers of AR in human sperm. To determine the efficacy of AR induction by glycopolymers, a flow cytometry assay is currently under development, which will allow for efficient sorting and quantitative analysis of dead, live, and acrosome-reacted cells. With the aid of this assay, promising AR inducers can be identified and will allow for the distinction of sperm that are able to participate in fertilization and those that are not. The development of this flow cytometry assay will ultimately provide the clinical community with a new method of assessing male infertility that can predict AR activation, thus improving current techniques used for diagnosis and treatment.
 Abubakar Lawal Mohammed
Abubakar Lawal Mohammed
M.Sc. Organic Chemistry, Mewar Univ., 2021. Chittorgarh, Rajasthan, India
B.Sc. Chemistry, Federal Univ. Dutse, 2018. Dutse, Jigawa, Nigeria
Program/Dates Supported by NIGMS: Chemistry/ 2023
Title: Pyridoxal and Nicotinamide-Inspired Reactions for the Development of Synthetic Organic and Chemical Biological Tools
My research focuses on the synthetic recapitulation of a paradigmatic activity of PLP, using the charge-transfer (CT) photochemistry of catalytically-generated formylpyridine-amino acid Schiff bases to enable synthetic photodecarboxylase reactivity for decarboxylative amino acid functionalization. This approach makes use of “Templated-Charge Transfer” (TCT) to enable photochemistry unavailable in an intermolecular sense, resulting in high degrees of organization and consequently selectivity. We wish to apply this approach in protecting group-free functionalization of singular amino acids as well as short peptides towards an end goal of achieving highly selective biofunctionalization.
 Ian Outhwaite
Ian Outhwaite
B.A. Williams College, 2017
Program/Dates Supported by NIGMS: Pharmacology/ 2022-2024
Title: Maximizing Selective Inhibition of Clinically Observed MET Mutants
Mesenchymal epithelial transition factor (c-MET/MET), a receptor tyrosine kinase, is an oncogenic driver in numerous tumor types, notably non-small cell lung cancer (NSCLC) and melanomas. On-target mutations drive resistance against small-molecule MET inhibitors. I aim to comprehensively profile the selectivity of clinically approved MET inhibitors against MET mutants, and determine combinations of these inhibitors that maximize on-target activity and reduce off-target effects.
This work will provide a rationale for targeted clinically approved tyrosine kinase inhibitor combinatorial therapies against MET mutants. Indicated combinations of clinically approved inhibitors will 1) maximize on-target activity, 2) reduce off-target effects, and 3) circumvent the development of tyrosine kinase inhibitor resistance. These inhibitor combinations may provide a direct translational benefit for patients with MET-driven cancers, or those in which MET signaling represents an escape mechanism following therapy against other targets (ex: EGFR, ALK).
 Steven Pak
Steven Pak
PharmD , St John’s University, 2016
Program/Dates Supported by NIGMS: Molecular and Cellular Pharmacology/ 2020-2022
Title: Virtual screening of small molecule inhibitors for mutated fibroblast growth factor receptor 2 in intrahepatic cholangiocarcinoma
My goal of my proposed research is to discover small molecule inhibitor for mutated fibroblast growth factors 2 in intrahepatic cholangiocarcinoma patients (ICC). ICC is a deadly liver cancer that includes very few treatment options. A large group of patients that presents all types of liver malignancies present ICC. Without adequate treatment, life expectancy after diagnosis is very short. Currently, there are no specific inhibitors for the mutational active FGFR2. Therefore, the goal of this project is to design small molecular candidates through virtual screening to bind to the binding site of the mutated FGF2.
By utilizing crystallographic structures of hyperactive FGFR2, structural models were prepared for docking simulations. Because mutated FGFR2 does not exist, additional models will need to be created for simulation. Then, large-scale virtual screening of commercially available compound libraries will be used to identify potential drug-like ligands. After virtual screening, results were ranked ordered via relevant scoring functions. Then, subsets of the most promising ligands will be purchased and tested for activity.
 Saigoutham Paturu
Saigoutham Paturu
MSc Medicinal Chemistry, University of Pune, India
Program/Dates Supported by NIGMS: Pharmacology/ 2022-2024
Title: Relation between the sigma factor RpoS and NosP-NahK signaling, and the downstream regulation of virulence, quorum sensing and biofilm in Pseudomonas aeruginosa.
Pseudomonas aeruginosa are Gram-negative bacteria that form biofilms, which aids in their pathogenicity by protecting them from antibiotics. P. aeruginosa is the causative agent for acute infections associated with burns, urinary tract infections, and chronic lung infections in cystic fibrosis patients. Biofilms are also responsible for hospital-acquired infections such as ventilator-associated pneumonia, a major problem during the SARS- CoV-2 pandemic due to increased ventilator use, especially affecting patients with low immunity. These biofilms are also hard to treat with current antibiotics because of their protective layer, however, one promising strategy to combat this problem is by using nitric oxide (NO) gas to promote biofilm dispersal, making the bacterial communities more responsive to drugs.
Previously, our lab discovered the proteins, NosP (Nitric Oxide Sensing Protein) and the co-cistronic associated histidine kinase (NahK) that recognize NO and leads to biofilm dispersal in P. aeruginosa. However, it is still unclear on the molecular mechanism behind the working of the NosP-NahK system and if it has any other functions in the bacteria. My goal is to investigate the potential interaction between the stationary phase sigma factor RpoS and NosP including the regulation of nosP-nahK transcription further influencing virulence, quorum sensing and biofilm, in P. aeruginosa.
 Lia Parkin
Lia Parkin
B.A., cum lade, Cell & Molecular Biology, Barnard College, Columbia University, 2018
Program/Dates Supported by NIGMS: Molecular Genetics and Microbiology / 2020-2021
Title:Towards understanding the lipid transport pathway LprG-Rv1410c in mycobacteria, and its contribution to outer membrane biogenesis, membrane permeability, and antibiotic susceptibility
Mycobacteria, including the human pathogen Mycobacterium tuberculosis (Mtb) that causes tuberculosis (TB), have a complex and impervious double membrane that distinguishes them from Gram negative and Gram positive bacteria. Unfortunately, Mtb infects one fourth of the global population. More disturbingly, new cases of Mtb are increasingly resistant to frontline therapies. By elucidating mycobacterial membrane biogenesis pathways, we can reveal new drug targets and identify proteins that control membrane permeability.
The Jessica Seeliger lab focuses on the LprG & Rv1410c proteins, which form an operon and likely work together to transport lipids to the outer membrane of mycobacteria, more specifically referred to as the mycomembrane. Loss of the LprG-Rv1410c operon leads to attenuation in vivo and a growth defect under nutrient limitation in vitro. We recently used a computational docking screen in search of commercially available small molecule inhibitors of LprG. We discovered molecules that inhibit mycobacterial growth in an LprG-dependent manner with micromolar affinity, indicating that LprG can be specifically inhibited. Additionally, we found that a single residue in the cavity of LprG mediated interaction with one of our most potent compounds. Overall, despite having a large and generally hydrophobic pocket, LprG can be realistically inhibited and rational drug design is feasible.
Understanding LprG-Rv1410c mediated lipid transport is also of interest because these proteins are predicted to be important for intrinsic resistance to multiple TB antibiotics. It has also been shown experimentally that the loss of the LprG-Rv1410c operon leads to increased susceptibility to certain antibiotics. Therefore, targeting or inhibiting LprG-Rv1410c may promote the efficacy of antibiotics that normally cannot cross the mycomembrane efficiently.
In future studies, we will determine how LprG & Rv1410c mechanistically mediate lipid
transport, in order to obtain a model of mycobacterial lipid transport to the mycomembrane.
Additionally, we will investigate how LprG & Rv1410c affect membrane permeability
in order to better understand drug diffusion across the mycomembrane. Ultimately,
this work will better define mechanisms of mycobacterial membrane biogenesis and drug
uptake. 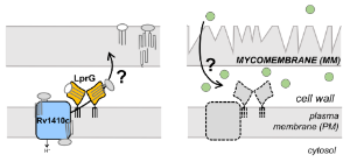
 Rideeta Raquib
Rideeta Raquib
B.S. in Interdisciplinary Biology, Stony Brook University
Program/Dates Supported by NIGMS: Biochemistry/ 2022- 2024
Title: Evaluating the Structure and Activity of Diacylglycerol-O-Acyltransferase 2 (DGAT2) in Various Lipid Membrane Environments
Triglycerides (TGs) are the main chemical form of long-term energy storage and are stored intracellularly in lipid droplets (LDs). TG accumulation is linked to various diseases including cancer, diabetes, cardiomyopathy, and obesity. LDs are generated in the endoplasmic reticulum (ER), where most of the enzymes involved in TG synthesis are located. This includes Diacylglycerol-O-Acyltransferase 2 (DGAT2), a monotopic membrane protein that catalyzes the last and only committed step of TG biosynthesis. Specifically, DGAT2 catalyzes the formation of an ester linkage between a fatty acyl-coA and 1,2-diacylglycerol (DAG), not only on the cytoplasmic face of the ER, but also on the surface of LDs, when fatty acids are in excess. Alternatively, DGAT2 can utilize pro-apoptotic ceramide as a substrate, instead of DAG, to generate 1-O-acylceramide, which has been implicated in causing cancer cells to become resistant to chemotherapy. Given DGAT2’s prominent roles, it serves as a strong candidate for therapeutic targeting. Although we have a general idea about the function and localization of DGAT2, little is known about its structure, the residues that facilitate selective substrate binding (e.g. DAG vs ceramide), and if catalytic activity is altered by diverse membrane environments (e.g. ER lipid bilayers vs. LD lipid monolayers). We plan to address these questions biochemically, using purified human DGAT2 and/or a thermophilic DGAT2 homolog from C. thermophilum (Ct DGAT2). We were able to detergent solubilize and purify recombinant human DGAT2 and Ct DGAT2. We developed enzymatic assays using both TLC and liquid chromatography-mass spectrometry (LC/MS) and confirmed purified Ct DGAT2 is catalytically active.
Further, we reconstituted the enzyme into various membrane environments, such as liposomes and artificial lipid droplets to mimic ER and LD membranes. Eventually, we plan to determine the structures of active DGAT2 alone and bound to its substrates and/or inhibitors using X-ray crystallography. Our hope is that in the future, this research will pave the way for the development and classification of potent DGAT2 inhibitors, with optimized mode of delivery to LDs and ER membranes, that can improve treatment efficacy for cancer, and other diseases associated with TG accumulation.
 Rachel White
Rachel White
BS in Biology, Pepperdine University
Program/Dates Supported by NIGMS: Molecular Genetics and Microbiology/ 2020-2022
Title: The Role of Host Protein TFII-I in Adenovirus Infection
For successful viral replication to occur, viruses must be able to subvert host cell immune mechanisms. In Adenoviruses, a family of nonenveloped DNA viruses, the Adenovirus early protein E4ORF3, a SUMO E3 ligase, has been shown to inhibit host responses that could otherwise block viral replication. Examples of targets with known functions include several involved in detecting and repairing double-strand DNA breaks, such as the Mre11-Rad50-Nbs1 (MRN) complex and p53. If not inhibited by the virus, these proteins can negatively impact Ad replication by responding to the virus’ double-stranded DNA genome as they would to a chromosomal double-strand DNA break. E4ORF3 also targets PML nuclear bodies, which are involved both in cell responses to DNA damage and in transcriptional regulation and antiviral responses such as the interferon response. Another major target of E4ORF3 is the transcription factor TFII-I, which suggests that it is involved in suppressing Ad infection. TFII-I is involved with multiple transcription pathways, including some activated by p53 in response to DNA damage, and can inhibit the transcription of the Ad protein L4, which is important for the transition between early and late viral transcription in Ad infection. Some studies have also associated TFII-I with the DNA Damage Response that the MRN complex functions in. However, its exact function in Ad infection is not known, and represents an important area of research to better understand the cellular mechanisms involved in Ad infection and possible general functions of TFII-I in viral infections.
Our lab has already created TFII-I CRISPR knockouts in normal human bronchial epithelial cells (NHBEC), and Ad strains that lack E4ORF3. I am also currently developing TFII-I CRISPR knockouts in small airway epithelial cells (SAEC). The goal of my project is to investigate cellular antiviral responses to Ad infection and Ad expression of early and late viral proteins both with and without TFII-I and E4ORF3 expression. Such investigation has the potential to increase our understanding of TFII-I as an anti-viral effector protein and of cellular mechanisms with the potential to block DNA viruses from successfully completing infection.
 Jason M. Withorn
Jason M. Withorn
BS Biochemistry and Chemical Biology from Wayne State University in Biology, Pepperdine University
Program/Dates Supported by NIGMS: Chemistry/ July 2021-present
Title: Functional characterization of the NosP associated histidine kinase, NahK, and its role in biofilm formation in Pseudomonas aeruginosa
Biofilms are multicellular communities formed when bacteria attach to a surface and secrete a thick, mucoid protective barrier known as the exopolysaccharide matrix (EPS). The EPS is composed of polysaccharides, extracellular DNA, and proteins. The EPS encompasses the cell promoting a nutrient rich environment that prevents antibiotics from reaching the cell.
It has been shown that the diatomic gas, nitric oxide (NO), facilitates the dispersal of biofilm-forming bacteria, reverting to a planktonic state. In 2017, our group discovered a novel heme-binding protein named NosP, nitric oxide sensing protein which functions to inhibit the NosP associated histidine kinase, NahK, which is located on the same operon. Our group has shown that when these two proteins are deleted, the bacteria are unable to form complex biofilm structures and they lose the ability to respond to toxic levels of NO. My project focuses on the pathogen Pseudomonas aeruginosa, which has been identified as a priority one pathogen needed a new treatment according to the World Health Organization. My project focuses on elucidating the function of NahK in vivo, and determining the role of the three uncharacterized PAS sensory domains located on the kinase. Understanding how NahK’s function in regards to signaling changes could allow for a more in depth understanding of NO-mediated biofilm dispersal, and allow for a more efficient treatment of biofilms to be developed.
The CBTP at SBU is made possible by a generous grant from the

